details
| Cardiac Marker | ||||||
| Catalog No. | Product Name | Specimen | Format | PCS | Sensitivity | CE |
| CTNI3002 | Cardiac Troponin I Rapid Test | S/P | Cassette | 25/40T | 0.5ng/ml | CE/NMPA |
| CTNI4002 | Cardiac Troponin I Rapid Test | WB/S/P | Cassette | 25/40T | 0.5ng/ml | CE/NMPA |
| CCK3002 | Creatine Kinase-MB Rapid Test | S/P | Cassette | 25/40T | 5ng/ml | CE |
| CCK4002 | Creatine Kinase-MB Rapid Test | WB/S/P | Cassette | 25/40T | 5ng/ml | CE |
| CMCT3312 | Myoglobin/CK-MB/Troponin I Tri-line Rapid Test | S/P | Cassette | 25/40T | 50/5/0.5ng/ml | CE |
| CMCT4312 | Myoglobin/CK-MB/Troponin I Tri-line Rapid Test | WB/S/P | Cassette | 25/40T | 50/5/0.5ng/ml | CE |
| CMYO3002 | Myoglobin Rapid Test | S/P | Cassette | 25/40T | 50ng/ml | CE |
| CMYO4002 | Myoglobin Rapid Test | WB/S/P | Cassette | 25/40T | 50ng/ml | CE |
Advantage
- 21 years experience
- Simple operation, reliable result
- High sensitivity and specificity,precision
- Cardic Marker test kits
Introduction
Product | TRO Rapid Test kit |
Specimen | Whole blood,plasma,serum |
Components | Individually packed test devices, Disposable pipettes, Buffer, Package insert |
Sensitivity | 100% |
Specificity | 99.8% |
Overall agreement | 99.8% |
Storage | 2-30℃ |
Validity | 24 Months |
Cardiac Troponin I (cTnI) is a protein found in cardiac muscle with a molecular weight of 22.5 kDa.1 Troponin I is part of a three subunit complex comprising of Troponin T and Troponin C. Along with tropomyosin, this structural complex forms the main component that regulates the calcium sensitive ATPase activity of actomyosin in striated skeletal and cardiac muscle.2 After cardiac injury occurs, Troponin I is released into the blood 4-6 hours after the onset of pain. The release pattern of cTnI is similar to CK-MB, but while CK-MB levels return to normal after 72 hours, Troponin I remains elevated for 6-10 days, thus providing for a longer window of detection for cardiac injury. The high specificity of cTnI measurements for the identification of myocardial damage has been demonstrated in conditions such as the perioperative period, after marathon runs, and blunt chest trauma.3 cTnI release has also been documented in cardiac conditions other than acute myocardial infarction (AMI) such as unstable angina, congestive heart failure, and ischemic damage due to coronary artery bypass surgery.4 Because of its high specificity and sensitivity in the myocardial tissue, Troponin I has recently become the most preferred biomarker for myocardial infarction.5
The Troponin I Rapid Test Device (Whole Blood/Serum/Plasma) is a simple test that utilizes a combination of anti cTnI antibody coated particles and capture reagent to selectively detect cTnI in whole blood, serum or plasma. The minimum detection level is 0.5 ng/mL.
Intend to use
The One Step TRO Test Device (Whole Blood/Serum/Plasma) is a rapid chromatographic immunoassay for the qualitative detection of human Myoglobin,
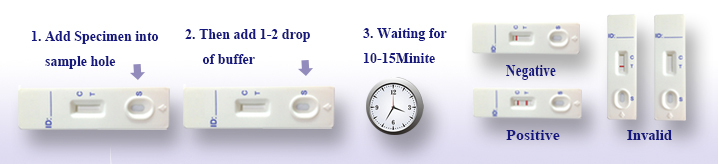
Operating Steps







评价